13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racem..

By A Mystery Man Writer
Solution For 13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g)
13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g) ₹ pressure. Then the ratio of the final volume to the initial volume of gases is:
Video solution 1: 13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g) ₹ pressure. Then the ratio of the final volume to the initial volume of gases is

Solved 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21

1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 24
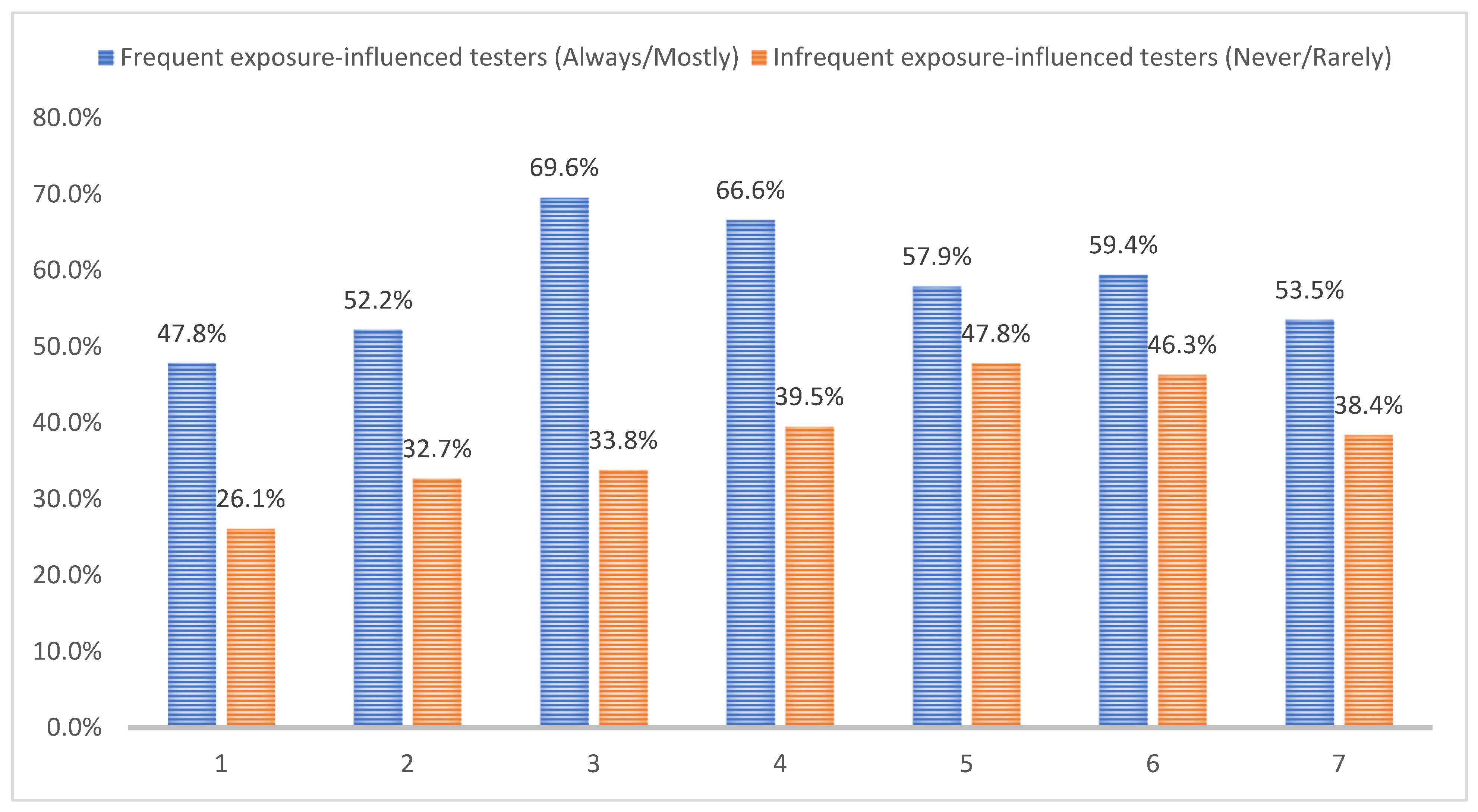
TropicalMed, Free Full-Text
Immigration to Portugal - Wikipedia
What is the next number in this sequence, 12, 13, 15, 18, ___? - Quora
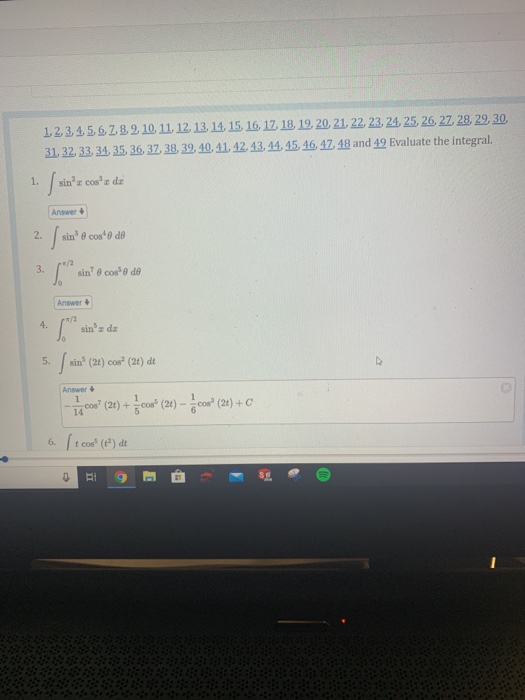
Solved 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15

Solved Consider the following data: 20 24 18 14 21 18 16 20
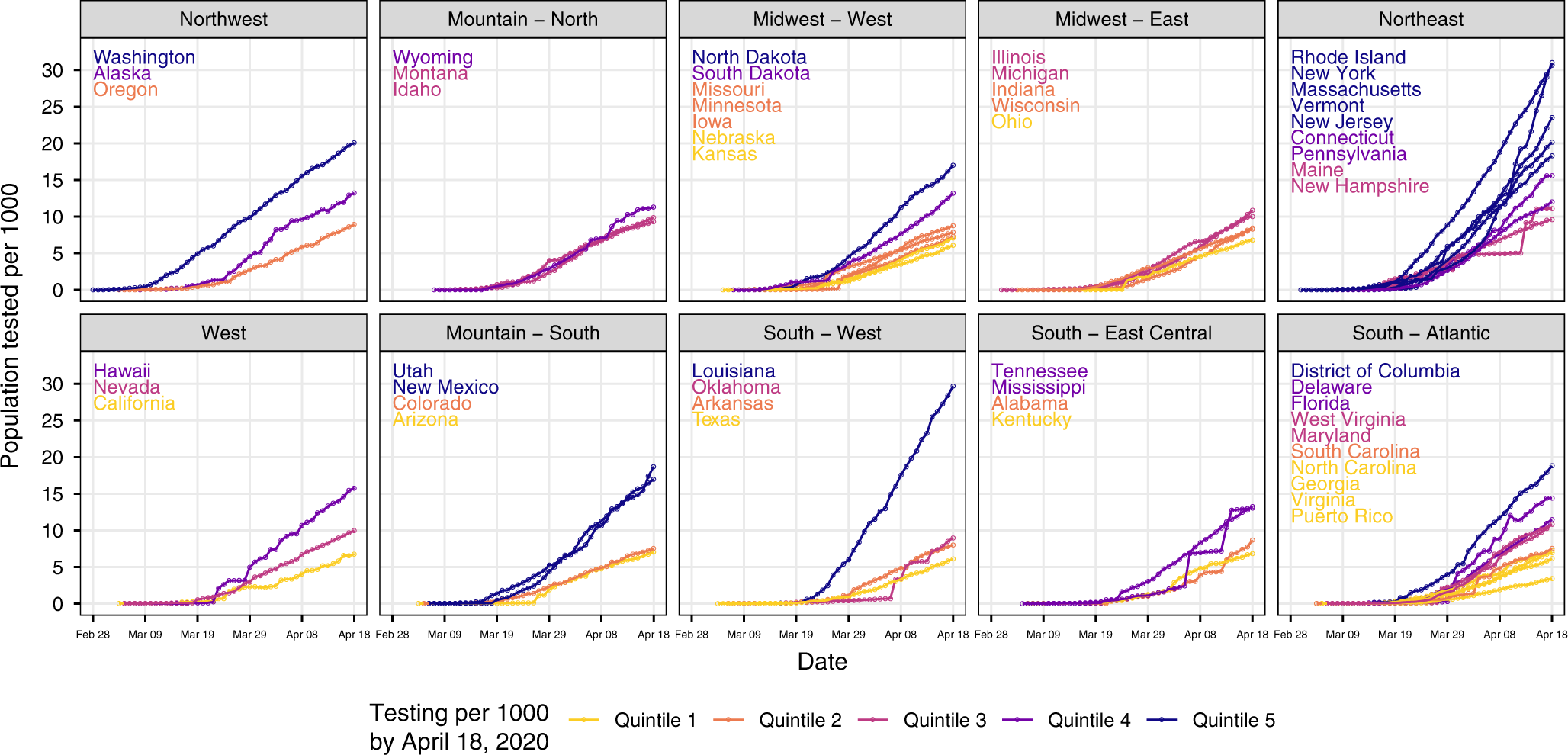
Substantial underestimation of SARS-CoV-2 infection in the United States
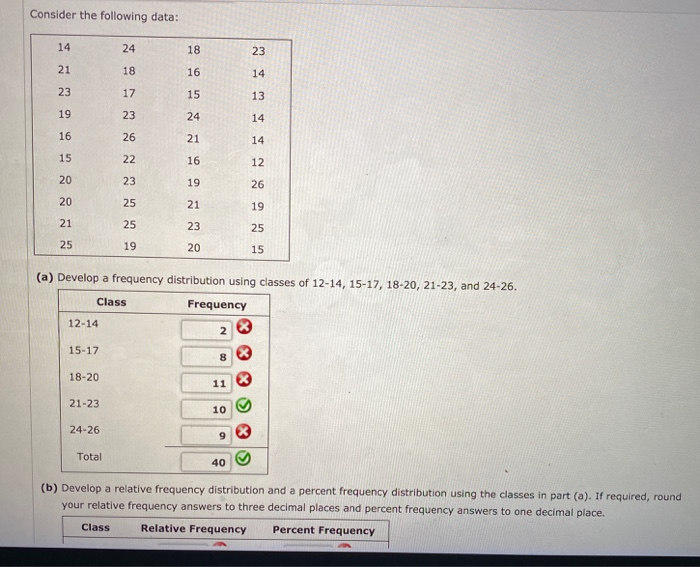
Solved Consider the following data: 14 24 18 23 21 18 16 14

13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of

Prova Fundação Hemocentro de BrasíliaDF - IADES - 2017 - para
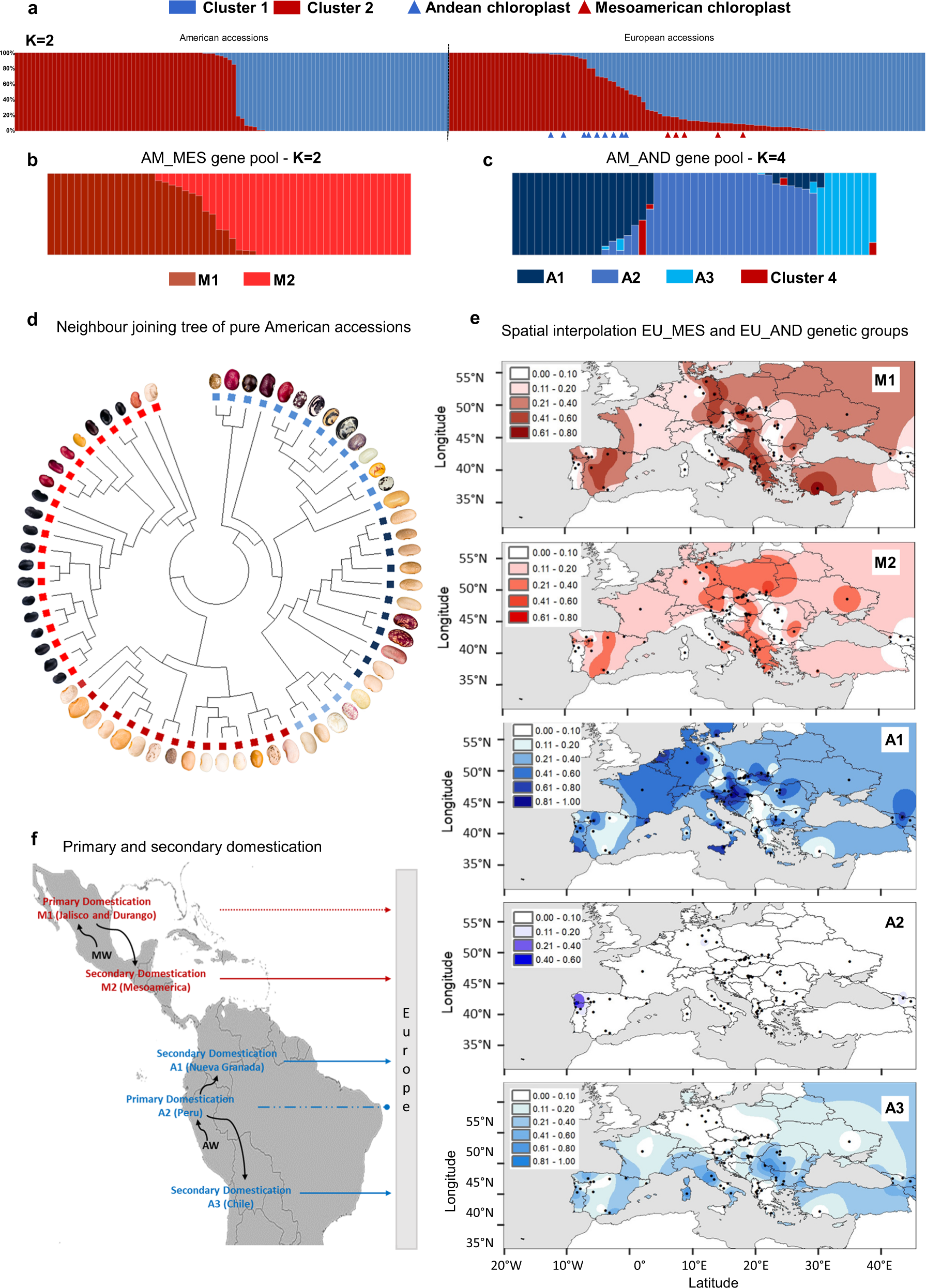
Selection and adaptive introgression guided the complex

ACADEMIC CATALOG - Luther Seminary
- What you need to know about the CPT and beyond…. WHAT is the College Placement Test (CPT)? The CPT measures skills in RD, WR and MTH These subjects. - ppt download

- VANITY FAIR Long Slip Nightgown - Size 34 32/40 Off White/ Beige - Made USA

- Higher temps mean higher food and other prices. Study links climate shocks to inflation

- An alloy contains 32% copper, 40% nickel and rest zinc. Find the mass of the zinc is

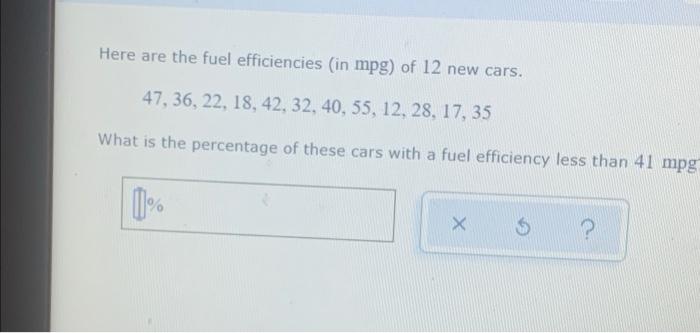
- Solved Here are the fuel efficiencies (in mpg) of 12 new
- Sports Bras for Women, Womens Cross Back Strappy Palestine

- Duo Ropa Interior Para Pareja Hello Kitty

- Unisex Full bodysuit Halloween Cosplay Men's Spandex Costume Suit Green Alien

- lululemon athletica, Pants & Jumpsuits, Lululemon All The Right Places Highrise Leggings Fuschia Luxtreme Sz

- Lace Up Adhesive Bra