Saturday, Sept 21 2024
Ideal Gas Assumptions - Kinetic Theory

By A Mystery Man Writer
When considering a gas as an ideal gas and applying the ideal gas law pV=nRT, we need to make 4 assumptions. (1) The volume of a molecule within the gas is n

PPT - Chapter 13 PowerPoint Presentation, free download - ID:4342474

Ideal Gas Law Formula and Examples

Molecular Motion of Gases

Kinetic Theory of Gases Formula, Assumptions & History - Physics
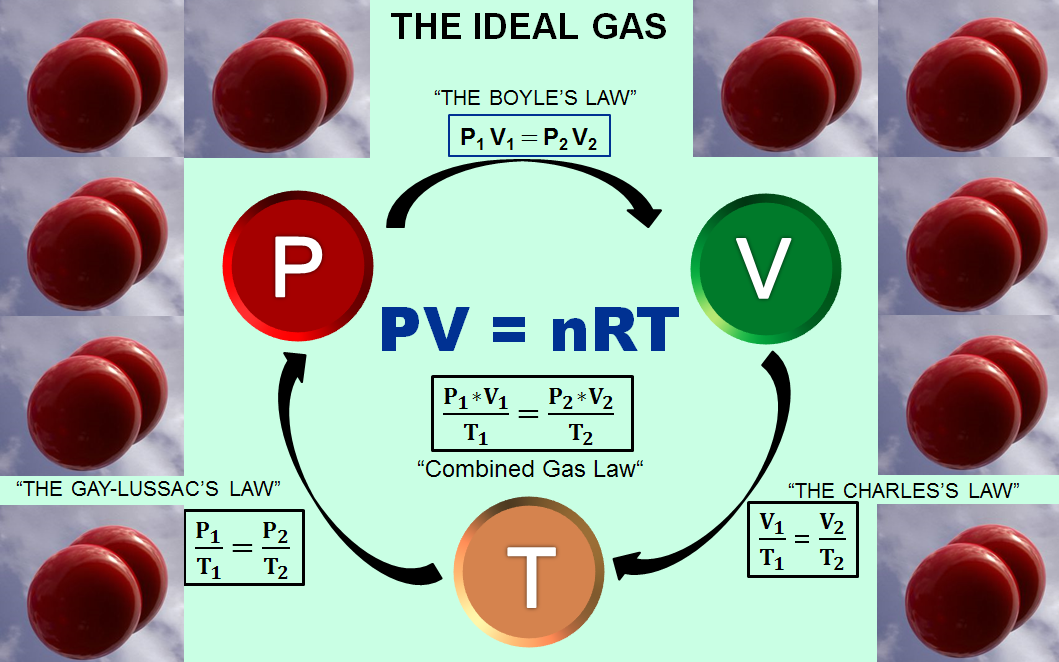
THE 3rd STATE OF MATTER – What is an Ideal Gas? – Computer Aided

Cameroon General Certificate of Education Compress, PDF, Heat
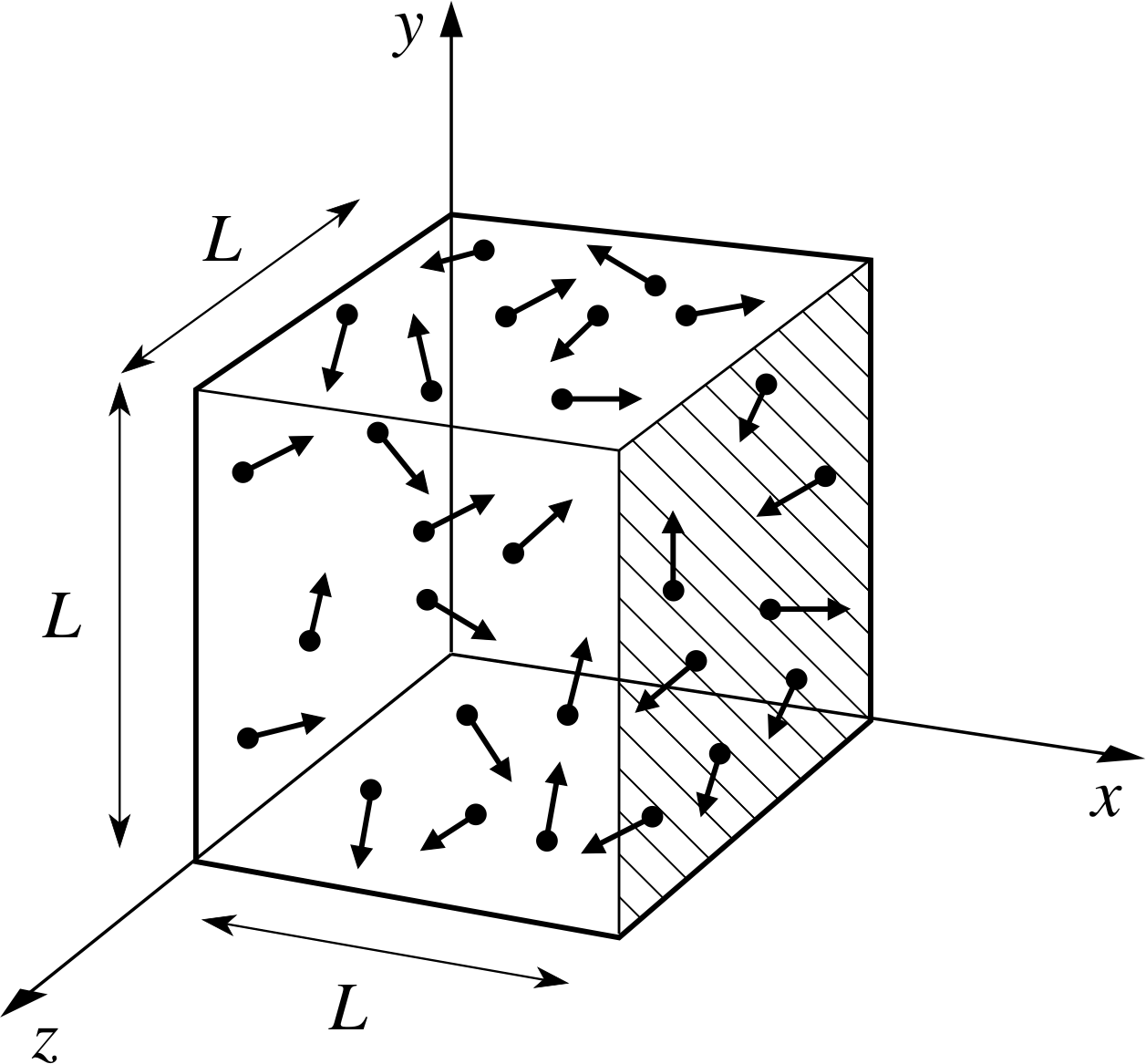
PPLATO, FLAP

ASSUMPTION OF IDEAL GASES

1-4 Gases (Part 3)

13.8 The Kinetic Molecular Theory of Gases - ChemistrySAANguyen
Related searches
- Ideal Gas Law: Statement, Characteristics, Formula & Problems
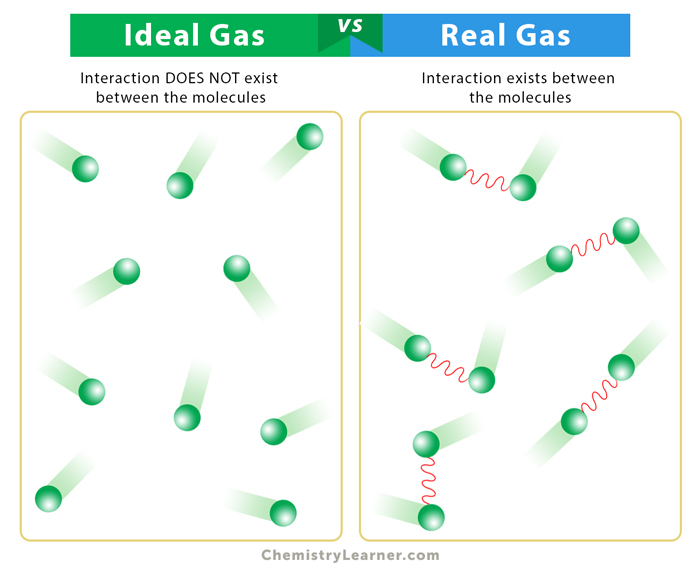
- 1,000+ Ideal Gas Stock Illustrations, Royalty-Free Vector Graphics & Clip Art - iStock

- Ideal Gas Law Brilliant Math & Science Wiki
- 10.4: The Ideal Gas Equation - Chemistry LibreTexts

- 6.5.3 Ideal Gas Equation, AQA A Level Physics Revision Notes 2017

©2016-2024, travellemur.com, Inc. or its affiliates





:extract_cover():upscale()/2018/03/21/646/n/44285655/289cf869c9be5c12_Everlane_1.jpg)